Eyewash solutions: Borate vs. Phosphate
Borate-based eyewashes are not equally effective, particularly against acids. Phosphate-based solutions remain the scientifically supported and safer choice for reliable eye protection.
Key difference: Buffering effectiveness
Phosphate-based eyewash has a strong and fast neutralizing effect on both acids and alkalis.
Borate-based eyewash, on the other hand, has shown limited neutralizing capacity, especially against acids.
Why phosphate matters
The eye’s natural pH is approximately 7.4. Phosphate buffers are physiologically compatible and help quickly restore the eye’s pH to this natural level. This reduces pain, tissue irritation, and longterm injury risk.
Clinical and safety implications
Borate-based eyewashes may appear similar but do not provide the same level of protection, particularly in industrial settings with acid exposure risks. Phosphate-based eyewashes (like ours) are therefore recommended by occupational safety specialists for comprehensive first aid readiness.
What this means for you
When advising your customers or replenishing first aid stations, choose phosphate-based eyewash to ensure:
- Optimal pH neutralization
- Proven effectiveness
- Compliance w/ modern safety standards
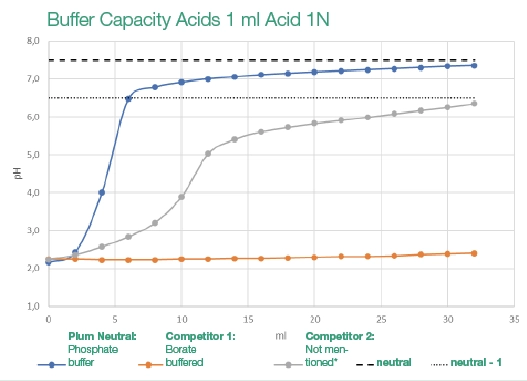
Testing by Labor Dr. Helle (2022), “Testing the buffer capacity of the eye rinsing solution,” demonstrated that borate solutions do not effectively stabilize pH levels after acidic exposure – a critical factor in preventing permanent eye damage.
*The ingredients and the quantitative composition are not mentioned by the manufacturer.
Important
Do not use this product for incidents with splashes containing hydrofluoric acid, calcium. or sodium hypochlorite (bleaches substanses).

Warning
Hydrofluoric acid is toxic and highly corrosive to eyes, skin, and bones. Plum Eyewash pH Neutral 4.9% Phosphate is not intended as first aid for eye accidents involving hydrofluoric acid, because it does not limit the corrosive effect of HF.

Warning
Substances containing calcium such as: calcium hydroxide, cement, and lime, may increase the risk of adverse effects if corneal damage occurs, which may lead to corneal calcification.

Warning
NaClO can be found in cleaning agents(bleaches). Although the irritating effect of alkali will be reduced by the use of buffer, the irritating effect of chlorine will be increased. Therefore, it is not recommended to use the product in case of such splashes.